Updated: April 2023
Published: July 2020
Languages: German, English
Based on the available scientific evidence and current clinical experience, the SPR Collaboration recommends that physicians and authorities consider the following covid treatment protocol for the prophylactic and early treatment of people at high risk or high exposure.
Numerous international studies have shown that prophylactic and early treatment can significantly reduce the risk of severe and fatal covid (see scientific references below).
Update: Covid treatments in retrospect (May 2024)
Note: Patients are asked to consult a doctor.
Treatment protocol
Prophylaxis
Goal: Supporting immune system, reducing viral load if infected.
- Vitamin D3 (2000 IU per day)
- Vitamin C (500mg per day)
- Zinc (20mg to 30mg per day)
- Quercetin (250mg per day)
- Mouthwashes and nasal spray
Early treatment
Goal: Preventing disease progression (anti-viral, anti-thrombotic, anti-inflammatory).
- Zinc (75mg to 100mg per day)
- Quercetin (500mg per day)
- Aspirin (325mg per day)*
- Bromhexine (48mg per day)*
- Arginine (3g to 5g per day)
- Artemisia annua (tea/powder)
- Mouthwashes and nasal spray
Prescription only
Goal: Preventing or reversing disease progression.
- High-dose calcifediol*
- Sulodexide (LMW heparin)
- Fluvoxamine or cyproheptadine*
- Steroids: Budesonide or prednisone*
- Ivermectin (see below)
Novel antiviral drugs
Goal: Targeted antiviral treatment.
- Paxlovid (Pfizer)
(*) Notes
- The early treatment phase includes post-exposure prophylaxis (PEP).
- Bromhexine is available prescription-free in most of Europe, but not in the US.
- Steroids and cyproheptadine are only used if respiratory symptoms develop.
- Counterindications and maximum dosages must be observed for all drugs.
- On hydroxychloroquine (HCQ), see Severe covid: A postviral autoimmune attack
See also
- FLCCC Covid-19 Prophylaxis and Treatment Protocols (FLCCC)
- Early Outpatient Treatment of COVID-19 (McCullough et al.)
- Covid-19 Early Treatment Study Overview (c19early.com)
General notes
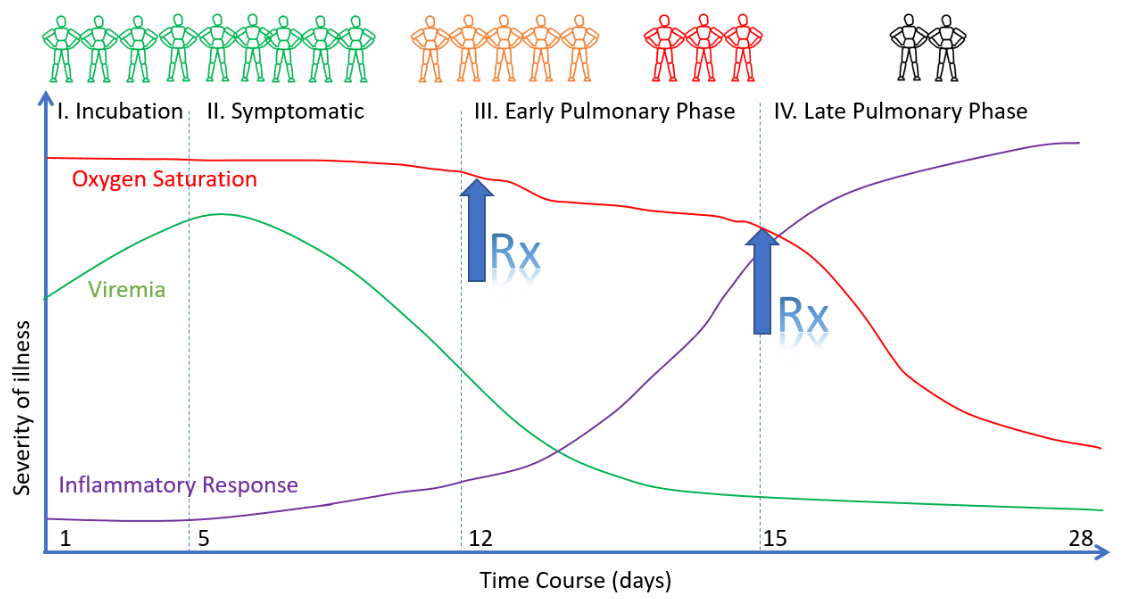
The early treatment of patients as soon as the first typical symptoms appear and even without a PCR test is essential to prevent progression of the disease. In contrast, isolating infected high-risk patients at home and without early treatment until they develop serious respiratory problems, as often happened during lockdowns, may be counterproductive.
People at high risk living in an epidemically active area should consider prophylactic treatment together with their doctor. The reason for this is the long incubation period of covid (up to 10 days): when patients first notice that they contracted the disease, the viral load is already at a maximum and there are often only a few days left to react with an early treatment intervention.
Early treatment based on the above protocol is intended to avoid hospitalization. If hospitalization nevertheless becomes necessary, it is recommended to avoid invasive ventilation (intubation) whenever possible and to use oxygen therapy (HFNC) instead.
Treatment studies
Results of trials and studies on the early treatment of covid.
Ivermectin
Ivermectin, an antiparasitic drug, has anti-viral and immuno-modulatory properties.
- Several high-quality trials of ivermectin found a non-significant advantage of about 20% to 30% concerning covid hospitalization and death in high-risk patients.
- Studies that claimed much higher effectiveness turned out to be unreliable.
- Professor Edward Mills, the lead investigator of the international TOGETHER trial, said that “there is a clear signal that ivermectin works in COVID patients” (April 2022).
- The renowned French Institute Pasteur found a “more favorable clinical presentation”, but not a reduced viral load, in a detailed coronavirus animal study.
- Some high-quality randomized trials of ivermectin are still ongoing.
- Read more: The Ivermectin Debate (SPR)
Zinc and quercetin
Zinc inhibits RNA polymerase activity of coronaviruses and thus inhibits virus replication. Quercetin (a plant polyphenol) supports the cellular absorption of zinc and has additional anti-viral properties.
- A Spanish study found that low plasma zinc levels (below 50mcg/dl) increased the risk of in-hospital death of covid patients by 130%.
- US studies found an 84% decrease in hospitalizations and a 45% decrease in mortality based on risk-stratified early treatment with zinc and HCQ.
- A US case study reported a rapid resolution of covid symptoms, such as shortness of breath, based on early outpatient treatment with high-dose zinc.
- An Italian randomized trial found a significantly reduced hospitalization rate and mortality in covid patients receiving quercetin.
Bromhexine
Bromhexine (a mucolytic cough medication) inhibits the expression of cellular TMPRSS2 protease and thus the entry of the virus into the cell, as first described in 2017.
- A randomized Iranian trial with 78 patients found a decrease in intensive care treatments of 82%, a decrease in intubations of 89%, and a decrease in deaths of 100%.
- Another Iranian study (randomized, double-blind, multi-center, preprint) with 370 contact persons found a 53% reduction in symptomatic confirmed covid.
- A Chinese trial found a 50% reduction in intubations in patients receiving bromhexine.
- A Russian study found a faster recovery in hospitalized patients receiving bromhexine.
- A Russian prophylaxis study found a reduction in symptomatic covid from 20% to 0%.
Vitamin D3
Vitamin D3 supports and improves the immune system response to infections.
- A Spanish randomized controlled trial found a 96% reduction in the risk of requiring intensive care in patients receiving high-dose vitamin D (100,000 IU).
- Another Spanish study with 930 hospitalized patients found a 87% reduction in ICU treatment and a 79% reduction in mortality in patients receiving high-dose vitamin D.
- A study in a French nursing home found an 89% decrease in mortality in residents who had received high-dose vitamin D either shortly before or during covid-19 disease.
- A retrospective British study of approximately 1000 hospitalized covid patients found an 80% reduction in mortality with high-dose vitamin D.
- A large Israeli study found a strong link between vitamin D deficiency and covid-19 severity.
- For an overview of all covid-19 vitamin D studies, see here.
Aspirin and heparin
Aspirin and heparin have anti-platelet and anti-thrombotic effects.
- A meta-study including 15,000 patients found a reduction in covid mortality of 53% in patients who were receiving aspirin as early or prophylactic treatment.
- A study published in PLOS One found a reduction in covid mortality at 30 days from 10.5% (control group) to 4.3% (with aspirin) in 70,000 US veterans taking aspirin.
- A retrospective US study with 400 patients found a reduction in ICU treatments by 43% and a reduction in mortality by 47% in the group of patients treated early with aspirin.
- A Mexican randomized controlled trial found a 40% reduction in hospitalization in patients receiving sulodexide (a heparin combination).
Arginine
Arginine is an amino acid supporting endothelial function, immune regulation, and tissue repair.
- A small randomized controlled trial, published in The Lancet E-Clinical Medicine, found that hospitalized covid patients receiving 3g of arginine per day had a significantly reduced need of respiratory support at 10 days and a significantly shorter length of hospitalization.
- A metabolomic analysis in covid ICU patients, published in Critical Care Explorations, found that low levels of arginine very strongly predicted the risk of death.
- A report published in PNAS found that plasma arginine and arginine bioavailability were significantly lower in adult and pediatric covid patients compared to controls.
- See also: l-Arginine and COVID-19: An Update (Nutrients, November 2021)
Artemisia annua
Artemisia annua plant extract has known antimalarial and antiviral properties.
- An in vitro study by the German Max Planck Institute, published in Nature Scientific Reports, found artemisia annua to be effective against SARS-CoV-2 at realistic doses.
- An in vitro study by US researchers, published in the Journal of Ethnopharmacology, found artemisia annua hot-water extracts to be effective against SARS-CoV-2 replication.
- In a small clinical trial, published in Antimicrobial Agents, the viral load decreased significantly faster in covid patients treated with artemisinin-piperaquine.
- In a 2005 in vitro study, published in Antiviral Research, artemisia annua was already identified as effective against the original SARS-1 virus.
- In Madagascar, the first country to apply artemisia annua extract against covid, covid mortality and excess mortality have remained very low, even for African standards.
- In a simulation study by a Harvard research group, the anti-malaria drug amodiaquine was identified as most effective against SARS-CoV-2 replication.
Mouthwashes and nasal sprays
Mouthwashes and nasal sprays target the initial infection and viral replication.
- Several small studies found that mouthwashes (gargling) based on povidone-iodine and nasal sprays based on povidone-iodine or nitric oxide may prevent a coronavirus infection or reduce its duration or symptoms (more about this).
- The German Society for Hospital Hygiene (DGKH) recommends anti-viral mouthwashes and nasal sprays for prophylaxis and early treatment.
- Some observers argued that traditional nasal rinsing and gargling practices in South East Asia may have helped successfully limiting coronavirus infections in these countries.
- Israel started mass production of nasal sprays based on nitric oxide in early 2021.
Fluvoxamine and Cyproheptadine
These drugs inhibit serotonin-induced pulmonary vasoconstriction.
- The TOGETHER trial found that fluvoxamine outpatient treatment of covid patients reduced disease progression (hospitalizations or ER visits) significantly by 32%.
- The results of two preliminary US trials indicate that early treatment with fluvoxamine may strongly reduce the risk of severe covid, hospitalization and death.
- In a small US case study, cyproheptadine rapidly improved the condition of hospitalized covid patients
- Fluvoxamine and cyproheptadine target serotonin metabolism and serotonin-induced pulmonary vasoconstriction, which according to emerging evidence may be a key mechanism driving severe covid and covid-related respiratory failure.
- Fluvoxamine is a selective serotonin reuptake inhibitor (SSRI) and reduces platelet serotonin concentration; cyproheptadine is a direct serotonin receptor antagonist. In addition, fluvoxamine is a sigma-1 receptor antagonist and has an anti-inflammatory effect.
Corticosteroids
Corticosteroids (e.g. prednisone, budesonide) reduce covid-related inflammation.
- A study by the University of Oxford, published in the Lancet, found a significant reduction in urgent care visits and hospitalizations in patients receiving budesonide (an asthma drug).
- The British PRINCIPLE trial found that budesonide accelerated recovery by 3 days and reduced hospitalizations and deaths from 10.3% to 8.5%.
- The early outpatient treatment protocol developed by McCullough et al. recommends the use of prednisone if (and only if) covid-related respiratory symptoms develop.
Anti-viral drugs
These novel drugs have direct anti-viral properties.
- Paxlovid (Pfizer) directly inhibits viral replication and, according to initial data, has been shown to be very effective in early treatment (reducing hospitalizations and deaths in at-risk groups by nearly 90%). Paxlovid, however, will not be available until mid-2022.
- Molnupiravir (Merck) has a questionable efficacy record. In addition, the mechanism of action is potentially DNA-toxic and may promote dangerous mutations of the virus. French health authorities rejected authorization of the drug.
- Monoclonal antibodies have been shown to be effective in early treatment but ineffective in the treatment of already hospitalized patients (post-viral phase). In addition, monoclonal antibodies rapidly lose efficacy against new viral variants.
- Plasma from convalescents has been shown to be ineffective in both early and late treatment of covid patients.
