Updated: April 2022
Published: July 2021
Share on: TG / TW / FB
Is ivermectin highly effective or totally useless against covid? And why is there still no agreement on this question?
Please note: Patients are asked to consult a doctor.
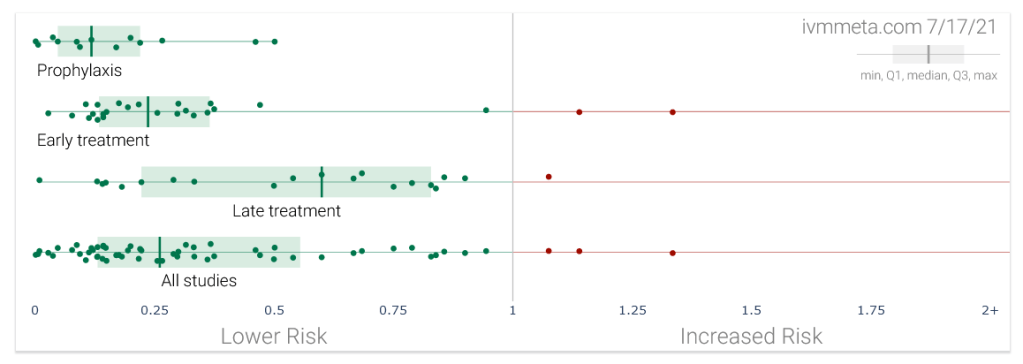
To date, about 60 studies – among them about 30 randomized controlled trials and about 40 peer-reviewed publications – have been done on the use of ivermectin against covid. Most of these studies found that the use of ivermectin was associated with a positive outcome, such as a reduced risk of infection, hospitalization or death (see chart above; not all of these results were significant).
Based on these studies, several meta-analyses computed positive results, too. Most recently, a WHO-supported meta-analysis of 24 randomized controlled trials found a 56% mortality reduction overall and a 70% mortality reduction in patients who received early outpatient treatment. There even appears to be a positive dose-response relationship, which is another indicator of effectiveness. (Update: The WHO-supported meta-analysis has been updated, see postscript below.)
Nevertheless, several concerns have been raised regarding the reliability of these results, and major – although not necessarily “independent” – health authorities like the WHO, the US FDA and the European EMA all continue to advise against the use of ivermectin outside of clinical trials.
First, it has been argued that there might be some publication bias, that is, only positive studies may have been published, while negative studies may have remained unpublished. However, the above mentioned meta-analysis was able to show that there is in fact no publication bias: studies that had been registered or announced did indeed get published, at least as a pre-print.
But secondly, and more importantly, it has been argued that many of the existing studies are of rather low quality: due to budget constraints, many of them are small, single-center, open-label (not double-blinded) and not perfectly randomized, thus significantly limiting their reliability. It is possible that at least some of these studies were biased towards getting a positive result.
Thirdly, and most worryingly, some of the ivermectin studies may simply be fraudulent or fake. As a matter of fact, one of the first positive studies on ivermectin, published in mid-April 2020, was from the very same group (Surgisphere) that published a fraudulent (negative) study on HCQ in the Lancet (both studies were ultimately retracted). And just last week, another seemingly positive study on ivermectin by an Egyptian group was shown to be very likely fraudulent. (See updates below.)
It remains debatable whether the groups behind these fraudulent studies simply tried to jump on the bandwagon of a promising medication, or if their role was in fact more substantial. In a context unrelated to ivermectin and covid, a former editor of the British Medical Journal recently argued that “we have now reached a point where those doing systematic reviews must start by assuming that a study is fraudulent until they can have some evidence to the contrary.”
Finally, and contrary to what some ivermectin proponents have argued, the epidemiological evidence in favor of ivermectin is rather weak. Ivermectin hasn’t “crushed the curve” anywhere; rather, decreases in infections were mostly driven by seasonal and endemic effects. In fact, ivermectin pioneers like Peru and Mexico have some of the highest covid mortality rates in the world; to their defense, even in Latin America, ivermectin use has often been limited and local.
On the other hand, the few studies claiming that ivermectin did not work against covid are also of rather low or dubious quality. The most famous one – a Colombian trial published in JAMA – was done so poorly that over 100 scientists and doctors called for its retraction. Furthermore, the only negative meta-analysis simply excluded most positive trials, confused the control and treatment group of another trial, and made several statistical mistakes, also triggering calls for retraction.
Some of the weakest arguments against ivermectin include questioning its safety (as done by Merck, the WHO and the US FDA, despite the fact that it is one of the safest drugs in the world), or calling it a “veterinary medicine” (it is used as an anti-parasitic drug in both humans and animals, as are many other drugs). Such dubious arguments, as well as some rather obvious conflicts of interest, raise legitimate questions about the motivations of some of the critics of low-cost ivermectin.
Regarding the potential mode(s) of action of ivermectin against Sars-CoV-2 and covid, proposals by various studies (molecular simulations, in vitro and in vivo) range from direct anti-viral action or interference with cellular receptors to immuno-modulatory mechanisms. Critics argue that doses needed to achieve anti-viral action are too high, but some ivermectin study authors have disagreed.
Most recently, a detailed covid animal study by the renowned French Institute Pasteur found that ivermectin “limited inflammation and prevented clinical deterioration”, but did not reduce viral load. The study “supports the use of ivermectin as an immuno-modulatory drug in covid patients”, but it would also, if applicable to humans, directly question the validity of several studies that claimed ivermectin works as an anti-viral prophylaxis against coronavirus infection (more on this).
To resolve this situation and finally answer the question of the effectiveness of ivermectin against covid, several high-quality randomized controlled trials (RCTs) are currently ongoing, sponsored by large foundations or public funds. While rather late in the pandemic, these trials are certainly of crucial importance to settle the ivermectin debate.
Large and expensive RCTs may, however, come with their own intricacies. Specifically, RCTs may to some extent be “designed to succeed” – as was the case with some covid vaccine trials as well as remdesivir – or be “designed to fail”. In the case of covid – a multi-phasic disease with a very steep age-based risk gradient – a trial can be designed to fail by enrolling (young) low-risk participants, using a drug late instead of early, under-dosing the drug or in some cases even over-dosing it.
For instance, the ongoing TOGETHER trial of ivermectin, sponsored by the Gates Foundation, was caught using just a single dose of ivermectin, whereas successful trials used two to four doses per day for up to five days. The Oxford PRINCIPLE trial, meanwhile, was caught enrolling participants up to 15 days after symptom onset, at which point some high-risk covid patients are already dead. Of note, the Oxford group had previously “botched” several other early treatment trials.
An even deeper issue, however, is that almost all covid medication trials have been looking for a single “wonder drug”, whereas it is increasingly clear that severe covid is an auto-immune, hyper-inflammatory and pro-thrombotic condition, which in high-risk patients may have to be treated by a combination of drugs (and as early as possible). This is becoming all the more important as the long-term protection by covid vaccines appears to be falling increasingly short of expectations.
Finally, ivermectin is also a prime example of the media war that has been going on during the covid pandemic, with US social media platforms – most notably Facebook and Youtube – having censored numerous doctors, scientists and politicians supporting ivermectin, while the GAVI vaccine alliance has been buying Google ads discouraging its use. Moreover, one of the major social media “fact checking” organizations turned out to have financial ties to a covid vaccine manufacturer.
In conclusion, the current evidence base concerning the use of ivermectin in the early treatment of covid continues to be positive, but important questions regarding the quality and certainty of many studies remain. It is to be hoped that ongoing high-quality RCTs will be able to resolve the debate. Given a still rising global covid mortality of currently about ten million people, if ivermectin is even just 10% effective against covid, its professional use could already have saved a million people.
Update October 2021
Several additional ivermectin trials turned out to be likely fraudulent (detailed discussion here). Almost all of these “trials” had been run by groups from Arab countries, which are known to produce a disproportionate share of fraudulent medical studies. Once low-quality studies are excluded from the analysis, the mortality benefit of ivermectin is no longer statistically significant.
The epidemiological evidence doesn’t support a strong effect of ivermectin, either: for instance, in the Indian state of Uttar Pradesh, known for its use of ivermectin, the total death count turned out to be 43 times higher than the official covid death toll.
Some beneficial immuno-modulatory effect – as found by the French Institute Pasteur – is still plausible, but the available evidence has become rather uncertain. Two high-quality RCTs of ivermectin are still ongoing, and ivermectin recently passed the “futility threshold” in both of these trials (i.e. >33% chance of benefit).
Update February 2022
A newly published ivermectin trial from Malaysia (I-TECH), while purporting to show no benefit of ivermectin, in fact showed a positive trend in favor of ivermectin: despite several design flaws, in-hospital mortality was about 70% lower in the ivermectin group (p=0.09). Read more.
Update April 2022
The long-awaited TOGETHER trial has finally been published. While the authors conclude that ivermectin failed, the data once again indicates that ivermectin may be moderately effective in the early treatment of high-risk patients: hospitalizations were 17% lower, mechanical ventilation was 23% lower, and deaths were 12% lower in the ivermectin group, and these differences were even more pronounced (but still not statistically significant) in people over 50. The probability of superiority of ivermectin compared to a placebo was calculated as about 80%.
Professor Edward Mills, the lead investigator of the trial, stated that “there is a clear signal that ivermectin works in COVID patients”. Read more.
